Learn About Allografts

Recombinants and Combination
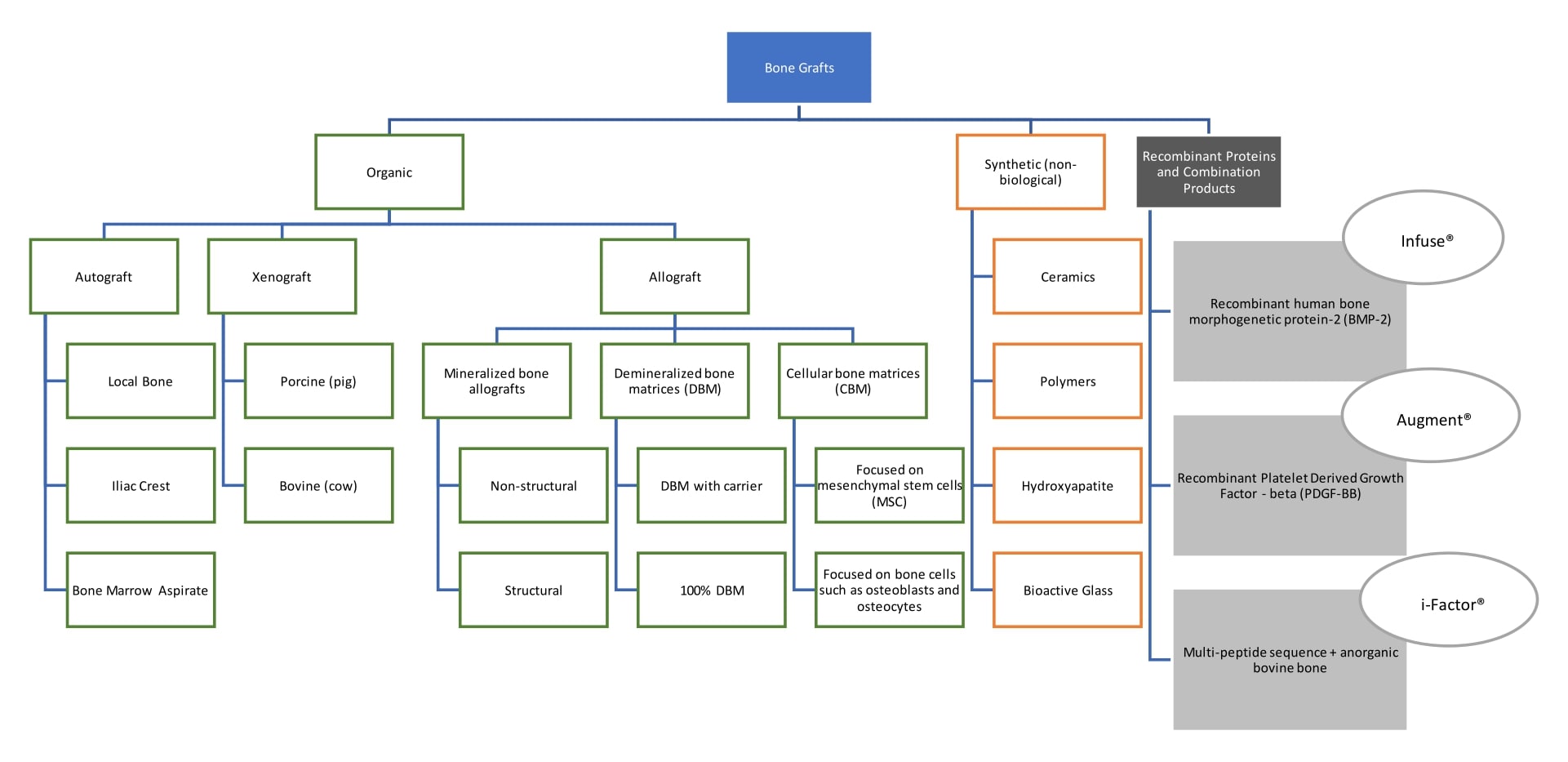
Bone grafting products with recombinant proteins provide a large dose of a signaling protein that is associated with bone formation. BMP-2 comes with a bovine collagen sponge which acts as a carrier for the growth factor.1 PDGF-BB is provided with beta tri-calcium phosphate that acts as a scaffold.2 Both of these products are considered combination products. There are several definitions of combination products, in for these, the relevant section "A product comprised of two or more regulated components, i.e., drug/device, biologic/device, drug/biologic, or drug/device/biologic, that are physically, chemically, or otherwise combined or mixed and produced as a single entity".3
An example of a combination product that does not include a recombinant protein is a short peptide sequence bound to anorganic bovine bone.
- Recombinant proteins or combination products that contain recombinant proteins are osteoinductive
- Depending on what other components are included, they may be osteoconductive
COMPARISON OF GRAFT PROPERTIES | |||
| Osteoconductive | Osteoinductive | Osteogenic | |
| Mineralized Allografts | |||
| Demineralized Allografts | |||
| Cellular Allografts | |||
| Autograft* | |||
| Synthetics | |||
Recombinant Proteins | |||
Combination Grafts | |||
| Xenograft | |||
*Autograft is partially demineralized, and therefore its osteoinductivity (signals) are unenhanced compared to grafts with fully demineralized bone.
- As Class III medical devices, recombinant proteins or combination products have specific indications listed in its Instructions for Use (IFU).
- All other use is considered off-label in contrast to allograft and autograft which can be used anywhere bone formation is needed.
- LifeNet Health offers natural bone allografts
References
- https://www.medtronic.com/us-en/healthcare-professionals/products/spinal-orthopaedic/bone-grafting/infuse-bone-graft.html Accessed 516.2022
- https://www.accessdata.fda.gov/cdrh_docs/pdf10/P100006d.pdf
- https://www.fda.gov/regulatory-information/search-fda-guidance-documents/classification-products-drugs-and-devices-and-additional-product-classification-issues. Accessed 5/13/2022